Apple has been instrumental in pairing medicine and mobile technology through its series of kits. In March 2015, they launched ResearchKit, an open source software framework to help scientists and doctors not only create medical apps but also recruit patients for clinical trials and research products.
ResearchKit turns iPhone into a powerful tool for medical research. When granted permission by the user, apps can access data from the Health app such as weight, blood pressure, glucose levels and asthma inhaler use, which are measured by third-party devices and apps. ResearchKit can also request from a user, access to the accelerometer, microphone, gyroscope and GPS sensors in iPhone to gain insight into a patient’s gait, motor impairment, fitness, speech and memory.
In March this year, they announced CareKit, a new development framework for clinical care apps. The software is designed help developers enable people to actively manage their own medical conditions through app-based care plans, and symptom and medication monitoring. This can leads to insights that help people better understand their own health.
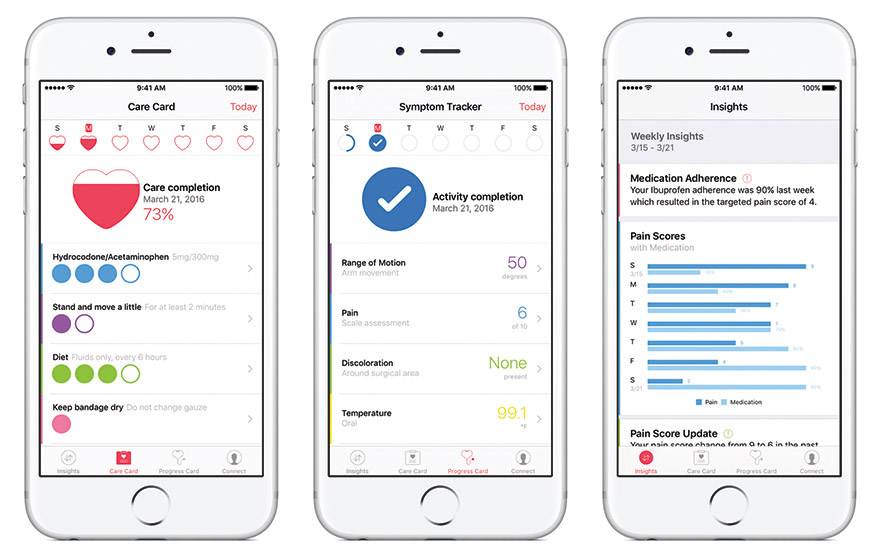
CareKit currently includes four modules:
- Care Card helps people track their individual care plans and action items, such as taking medication or completing physical therapy exercises. Activities can automatically be tracked and entered using sensors in Apple Watch® or iPhone
- Symptom and Measurement Tracker lets users easily record their symptoms and how they’re feeling, like monitoring temperature for possible infections or measuring pain or fatigue. Progress updates could include simple surveys, photos that capture the progression of a wound or activities calculated by using the iPhone’s accelerometer and gyroscope, like quantifying range of motion;
- Insight Dashboard maps symptoms against the action items in the Care Card to easily show how treatments are working
- Connect makes it easy for people to share information and communicate with doctors, care teams or family members about their health and any change in condition
Research kits has been met with positive results from the medical community. “With ResearchKit, we quickly realized the power of mobile apps for running inexpensive, high-quality clinical studies with unprecedented reach,” said Ray Dorsey, MD, David M. Levy Professor of Neurology at the University of Rochester Medical Center.
“We hope that CareKit will help us close the gap between our research findings and how we care for our Parkinson’s patients day-to-day. It’s opening up a whole new opportunity for the democratization of research and medicine.”
Apple seeks to professionalize health apps
Apple’s efforts leads to a greater professionalisation of ‘health and wellbeing’ apps- a lucrative industry that lacks any accredited regulation. At present they do not need to be designed by or in collaboration with health professionals or endorse tools or treatments that are subject to peer reviewed papers and research based trials. For example, a study released in 2015 analyzing 243 depression apps available during 2013 revealed that many fail to incorporate evidence-based practices, health behavior theory, or clinical expertise into their design. The study excluded almost this many again for the lack of reporting of organizational affiliation and content sources.
Likewise, a study was released this year of apps that explicitly included or advertised pregnancy prevention or decision-making support. Intensive analysis was undertaken of 18 apps. Of these, 41% of apps did not mention any modern contraceptive methods and 23% mentioned only 1 method. Of apps that did mention a modern contraceptive method, fewer than 50% of these apps provided information on how to use it. The researchers further found that most apps miss opportunities to provide users with valuable information, interactive decision aids, and evidence-based interventions for unintended pregnancy prevention. The researcher further contend that
“some apps in this space may increase the likelihood of unintended pregnancy due to the low effectiveness of the contraceptive methods promoted.”
Other research highlights the problem of clinical assessment of apps due to search terms/procedures used, the retail stores/research databases searched and the criteria that is used to determine app quality.
What is and isn’t a health app?
According to guidelines by the U.S. Food and Drug Administration (FDA) in February 2015, apps that are considered a medical device category, require FDA review before they can go to market legally. A medical device can include software that is intended for use in one of three situations:
- To diagnose a disease or other condition
- Cure, mitigate, prevent, or treat a disease
- Affect a bodily function or structure
Interestingly, there are also several categories of mobile apps for which the FDA plans to exercise enforcement discretion but not actively regulate. These include apps that:
- Help users self-manage their disease or condition without providing specific treatment or treatment suggestions
- Provide patients with simple tools to organize and track their health information
- Provide easy access to information related to a patient’s health conditions or treatments
- Help patients document, show, or communicate potential medical conditions to healthcare providers
- Automate simple tasks for healthcare providers
- Enable patients or providers to interact with personal health records or electronic health record systems
It’s highly likely that most developers of health apps who are not science or health professionals themselves, are most likely to want to avoid a FDA review-a process that is time consuming and expensive. Rather than reducing the amount of apps on offer, it is more likely to result in more generic or generalist knowledge or a self-classification under “well-being,” a category that the FDA considers most relative to a healthy lifestyle than health.
What is missing from AppleKits?
It’s notable that while Apple has been working hard to create software platforms suitable for iPhone apps, there has been something of a missed opportunity for engagement with other connected devices utilized now (or in the future) by health professionals that would utilize CareKit. For example, whilst patients can share medical data with doctors, there is no corresponding platform for integrating the data into patient electronic health records or a means to compare the data with other patients utilizing the same app. Whilst patient adherence is a common challenge of any health regime, without tools to ensure health professionals can also do so with the resultant data is imperative.